How Omeicos Identified Biomarker Signals and Safety Trends with Rivia
Background
Omeicos Therapeutics is a clinical-stage biotechnology company developing first-in-class therapeutics for cardiovascular and inflammatory diseases. Its lead compound, OMT-28, is a stable synthetic analog of an omega-3 metabolite, and is designed to reduce mitochondrial dysfunction and oxidative stress.
The company is conducting a Phase 2a evaluating OMT-28 in patients with Primary Mitochondrial Disease (PMD), a complex multi-system disorder. The trial required robust assessment of safety and efficacy, as well as an understanding of clinical response patterns and key biomarkers including GDF-15 and IL-6.
Rivia spoke with Robert Fischer (CEO/CSO and Co-Founder) and Henk Streefkerk (Drug Safety Officer) who oversee the scientific leadership, safety management, and overall data oversight of Omeicos’ clinical programs. Robert highlights Rivia’s impact throughout the study:
"Rivia has supported us across the entire trial lifecycle, from real-time patient monitoring to in-depth post-study analysis, enabling faster and more informed data-driven decisions"
— Robert Fischer, CEO/CSO and Co-Founder
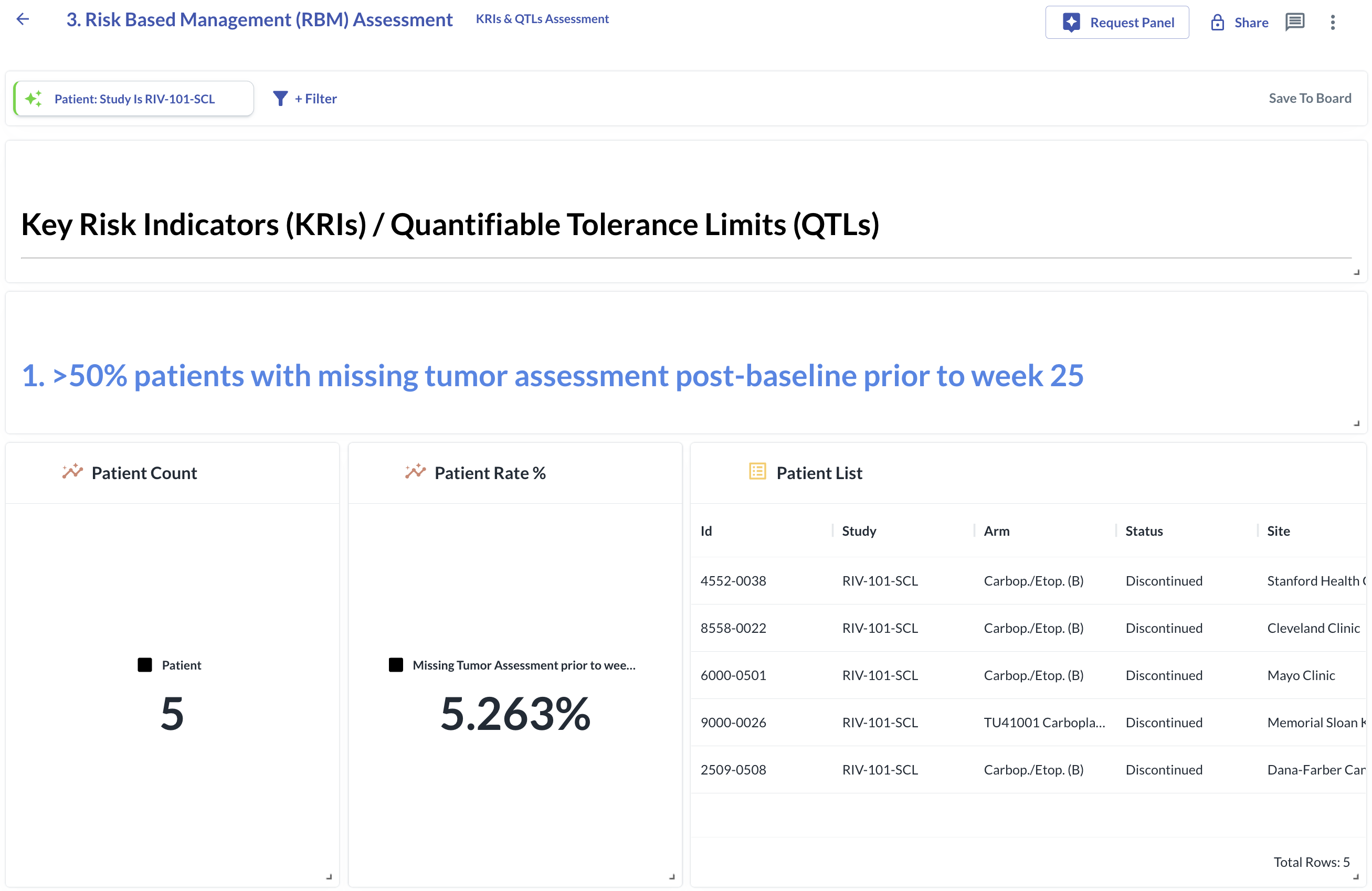
Challenges: Fragmented Data Slowed Critical Insight
Successfully managing the PMD-OPTION trial required handling large volumes of diverse patient data. Specifically, Omeicos faced three key challenges:
- Fragmented Data Across Multiple Sources - Data from the EDC, central laboratory, pharmacokinetics (PK), and physical tests existed across disconnected systems, making cross-comparison slow, error-prone, and limiting access to a unified patient view.
- Need for Real-Time Safety Monitoring - Correlating laboratory and functional test results over time for early efficacy and safety review was slow due to manual data consolidation, risking missed clinically meaningful developments.
- Subgroup Analysis - Identifying responder vs. non-responder groups post-study was hindered by fragmented datasets and the lack of an integrated analytical environment.
Solution: Unifying Complex Trial Data with Rivia
Omeicos adopted the Rivia data integration and analytics platform to consolidate all study data into a single environment. During the trial, Rivia provided the clinical team with a centralized platform for operational monitoring, medical oversight, and safety tracking. After study completion, Rivia enabled efficient subgroup exploration and endpoint analysis, allowing a seamless transition from day-to-day trial management to in-depth scientific interpretation.
1. Unified, Real-Time Patient Data for Medical and Safety Monitoring
Rivia connected all critical data sources into a comprehensive, real-time platform, making patient profiles accessible in seconds instead of hours or days.
“Rivia connected our various data sources - eCRF, lab results, and physical tests. This allowed us to quickly obtain a comprehensive view of every patient.
It allows us to quickly understand how our drug impacts patients, explore correlations, and uncover trends.”
— Robert Fischer, CEO/CSO and Co-Founder
“Rivia’s integrated safety workflows enable me to complete monthly reporting in minutes that previously would take half a day. Just as importantly, they help me detect signals I would have otherwise missed, allowing faster corrective actions and ensuring continuous safety profiling.”
— Dr. Henk Streefkerk, Drug Safety Officer
This real-time view enabled the team to monitor trends, compare endpoints, and assess lab and biomarker changes as the study progressed, shifting from reactive to proactive safety and efficacy management.
2. Accelerated Scientific Insight and Cohort Analysis
After study completion, Rivia became a key tool for scientific interpretation. The platform allowed rapid definition and analysis of patient cohorts, biomarker trends, and responder vs. non-responder characteristics. For instance, it enabled seamless exploration of subgroup differences in clinical outcomes, such as the Newcastle Mitochondrial Disease Adult Scale (NMDAS) scores.
“With Rivia, we can easily analyze patient cohorts and identify responder groups, giving us critical insight into why some patients benefit from our treatment while others don’t.”
— Robert Fischer, CEO/CSO and Co-Founder
These insights informed future development planning and supported the optimization of the clinical strategy for OMT-28.
3. Unified Data Throughout the Trial
Rivia harmonized all study data in a single platform from the first patient visit through final analysis. This continuity eliminated the need to switch between disconnected tools or rely on manual aggregation. During active treatment phases, Rivia delivered real-time visibility into patient status and safety signals, and post-study, it enabled rapid scientific exploration, subgroup discovery, and endpoint evaluation. Every operational and scientific decision was based on complete, integrated, and up-to-date study data.
Featured Testimonials
Read More
Latest News
.svg)







.png)

.svg)



